Print this page (or the pdf version using the link below) and answer each item before using the ChemLab courseware. Ask a teacher to correct your answers and report your score to you.
- PART I - Chemical Names, Symbols and Formulas
1. Water is the common name of the chemical compound H20.
True or False
2. Iron is one of the known naturally occurring elements.
True or False
3. Which of the following chemicals is a mixture?
water *** air *** helium *** vinegar
4. Hydrogen is:
an element *** a compound *** a mixture?
5. Sodium chloride is more commonly known as
______________
6. What is the chemical symbol or formula for Hydrogen?
_____
- PART II - Periodic Table Basic Information
7. Elements in the Periodic Table are arranged by common properties and behaviors.
True or False
8. The properties of the compound Sodium Chloride (NaCl) are the same as the properties of the elements Sodium (Na) and Chlorine (Cl).
True or False
9. What chemical information can be found on the Periodic Table?
atomic weight *** melting temperature *** physical state *** all of these
10. Which of the following would NOT be found on the Periodic Table?
gold *** steel *** mercury *** aluminum
11. Acetic Acid (CH3COOH) is comprised of which elements?
__________________
12. Inert gasses are arranged in what part of the Periodic Table?
_________________
- PART III - Chemical Reactions, Reactants and Products
13. A liquid that changes to a gas when heated is an example of a chemical reaction.
True or False
14. When Vinegar (acetic acid - CH3COOH) reacts with baking soda (sodium bicarbonate - NaHCO3), one of the products is Carbon Dioxide (CO2).
True or False
15. Indicators that a chemical reaction may have occurred include:
heat is produced *** formation of new substances *** gas is produced *** all of these
16. If sodium chloride (NaCl) is mixed with nitric acid (HNO3) what are the likely products?
calcium chloride (CaCl2) *** carbon dioxide (CO2) *** sodium hydroxide (NaOH) *** none of these
17. In question #16, what are the reactants?
_________________________________
18. Write a chemical reaction equation for the following: Water added to Iron
_______________________________________
- PART IV - Lab Equipment
19. Name the item in the picture
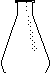 ___________________
___________________
20. Name the item in the picture
 ___________________
___________________
- PART V - Describe a chemical reaction situation or scenario
Include the reactants, expected products, and reaction indicators.
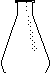 ___________________
___________________ ___________________
___________________